 Preclinical Assets
Preclinical AssetsPreclinical Assets Decade Long Trends, Analysis and Forecast 2025-2033
Preclinical Assets by Type (Bioanalysis and DMPK Studies, Toxicology Testing, Compound Management, Safety Pharmacology, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2025-2033
Key Insights
Market Overview and Drivers:
The global preclinical assets market is projected to grow significantly over the forecast period, driven by several factors. The rising demand for new drug development and the increasing adoption of preclinical testing services in the pharmaceutical industry are major contributors to this growth. Additionally, advancements in technology and the emergence of innovative techniques, such as bioanalysis and DMPK studies, are fueling market expansion. The market is segmented into types, including bioanalysis and DMPK studies, toxicology testing, and compound management.
Competitive Landscape and Regional Outlook:
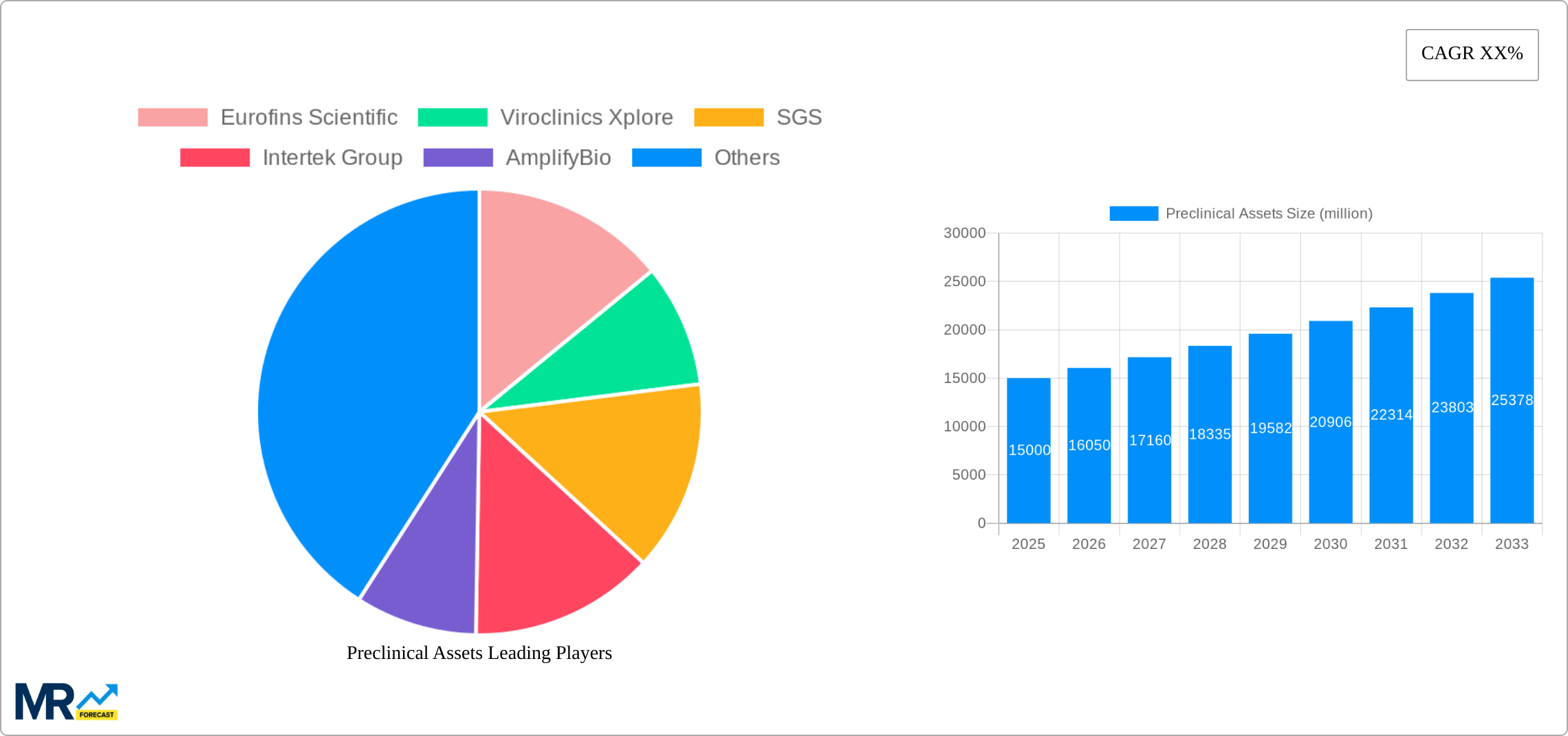
Key players in the preclinical assets market include Eurofins Scientific, Viroclinics Xplore, SGS, Intertek Group, AmplifyBio, and IQVIA. These companies offer a wide range of preclinical testing services and are actively involved in research and development to enhance their offerings. Geographically, North America and Europe dominate the market, owing to the presence of well-established pharmaceutical and biotechnology industries. The Asia-Pacific region is expected to witness substantial growth in the coming years due to the increasing investment in healthcare infrastructure and the growing pharmaceutical sector in the region.
Preclinical Assets Trends
The preclinical assets market is experiencing significant growth, driven by rising demand for novel drug therapies and increased outsourcing of preclinical research services. The market is valued at USD 19.3 billion in 2023 and is projected to reach USD 30.3 billion by 2028, exhibiting a compound annual growth rate (CAGR) of 7.3% during the forecast period. The increasing prevalence of chronic diseases, technological advancements, and government initiatives supporting drug development contribute to the market's expansion.
Key Market Insights
- Outsourcing of preclinical services is accelerating, driven by pharmaceutical companies seeking cost-effective and efficient solutions.
- Biotechnology and pharmaceutical companies are investing heavily in preclinical research to identify and validate potential drug candidates.
- Advanced technologies, such as high-throughput screening and bioinformatics, are transforming preclinical research processes, enabling faster and more accurate data analysis.
- Regulatory bodies emphasize the need for robust preclinical data, leading to increased demand for specialized preclinical testing services.
- Emerging markets offer growth opportunities for preclinical service providers, as they demand access to specialized expertise and infrastructure.
Driving Forces: What's Propelling the Preclinical Assets
The preclinical assets market is propelled by several key factors:
- Rising Drug Discovery Costs: The escalating costs of drug discovery and development drive pharmaceutical companies to outsource preclinical research to specialized service providers, reducing overheads and accelerating timelines.
- Technological Advancements: Technological innovations in areas like genomics, proteomics, and bioinformatics enhance the precision and efficiency of preclinical testing, leading to more informed decision-making.
- Increased Outsourcing: The growing trend of outsourcing preclinical services allows pharmaceutical companies to focus on core competencies and leverage the expertise of specialized providers.
- Regulatory Compliance: Stringent regulatory requirements demand robust preclinical data, driving demand for comprehensive safety and efficacy studies before clinical trials.
- Accelerated Drug Development: The need for faster drug development timelines drives the demand for preclinical services that can identify and validate potential drug candidates efficiently.

Challenges and Restraints in Preclinical Assets
The preclinical assets market also faces some challenges:
- Technological Complexity: Preclinical research involves complex technologies and methodologies, requiring specialized expertise and infrastructure, which may pose a challenge for smaller companies.
- Data Management: The generation of vast amounts of data in preclinical research poses challenges in data management, analysis, and interpretation.
- Regulatory Hurdles: Regulatory requirements can be complex and vary across jurisdictions, requiring careful navigation and compliance to ensure the validity of preclinical data.
- Ethical Considerations: Preclinical research involving animal models raises ethical concerns regarding animal welfare and the responsible use of animals in scientific experiments.
- Cost and Time Constraints: Preclinical research can be time-consuming and expensive, requiring careful planning and resource allocation to achieve desired outcomes within budget and timelines.
Key Region or Country & Segment to Dominate the Market
Key Region
North America and Europe are expected to dominate the preclinical assets market throughout the forecast period. These regions have well-established pharmaceutical and biotechnology industries, strong research infrastructure, and a supportive regulatory environment.
Key Segment
Safety Pharmacology is projected to dominate the preclinical assets market over the next five years. The increasing focus on drug safety and regulatory requirements for comprehensive safety assessments drive the demand for specialized safety pharmacology testing services.
Growth Catalysts in Preclinical Assets Industry
- Personalized Medicine: The rise of personalized medicine and precision medicine approaches requires tailored preclinical testing to assess drug efficacy and safety in specific patient populations.
- Artificial Intelligence (AI): AI-powered technologies are transforming preclinical research, enabling faster and more accurate data analysis, prediction of drug properties, and optimization of experimental design.
- Microfluidics: Microfluidics technology offers innovative methods for preclinical testing, reducing costs, improving throughput, and enabling more precise analysis of drug interactions and biological processes.
- 3D Cell Culture: Advanced 3D cell culture systems mimic organ-specific microenvironments, enhancing the physiological relevance and predictive power of preclinical research.
- Alternative Testing Models: The development of alternative testing models, such as organ-on-a-chip and computational modeling, provides more ethical and cost-effective approaches to preclinical research.
Leading Players in the Preclinical Assets
- Eurofins Scientific
- Viroclinics Xplore
- SGS
- Intertek Group
- AmplifyBio
- IQVIA
- ICON plc
- Medpace
- Pharmatest Services
- PPD
- Crown Bioscience
- Comparative Biosciences
- TCG Lifesciences
- Domainex
- Absorption Systems
Significant Developments in Preclinical Assets Sector
- Acquisition of Certara by EQT Private Equity: This acquisition strengthened EQT's capabilities in preclinical modeling and simulation, enhancing drug development efficiency.
- Expansion of Eurofins' Preclinical Services: Eurofins acquired several preclinical testing facilities, expanding its global footprint and service offerings.
- Collaboration between IQVIA and Biogen: IQVIA and Biogen partnered to develop innovative preclinical models for neurodegenerative diseases.
- FDA Guidance on Preclinical Safety Testing: The FDA issued updated guidance on preclinical safety testing, emphasizing the importance of robust and comprehensive data.
- Investment in Alternative Testing Models: Several companies are investing in developing organ-on-a-chip and other alternative testing models to reduce animal use and improve preclinical research accuracy.
Comprehensive Coverage Preclinical Assets Report
This report provides a comprehensive analysis of the preclinical assets market, covering key trends, driving forces, challenges, growth catalysts, and significant developments. It offers insights into the competitive landscape, including leading players and their strategies. The report also includes detailed market segmentation and regional analysis, providing valuable information for stakeholders in the preclinical research industry.
Preclinical Assets Segmentation
-
1. Type
- 1.1. Bioanalysis and DMPK Studies
- 1.2. Toxicology Testing
- 1.3. Compound Management
- 1.4. Safety Pharmacology
- 1.5. Others
Preclinical Assets Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific

Preclinical Assets REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2019-2033 |
| Base Year | 2024 |
| Estimated Year | 2025 |
| Forecast Period | 2025-2033 |
| Historical Period | 2019-2024 |
| Growth Rate | CAGR of XX% from 2019-2033 |
| Segmentation |
|
Frequently Asked Questions
How can I stay updated on further developments or reports in the Preclinical Assets?
To stay informed about further developments, trends, and reports in the Preclinical Assets, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Can you provide examples of recent developments in the market?
undefined
What is the projected Compound Annual Growth Rate (CAGR) of the Preclinical Assets ?
The projected CAGR is approximately XX%.
What are the notable trends driving market growth?
.
Which companies are prominent players in the Preclinical Assets?
Key companies in the market include Eurofins Scientific,Viroclinics Xplore,SGS,Intertek Group,AmplifyBio,IQVIA,ICON plc,Medpace,Pharmatest Services,PPD,Crown Bioscience,Comparative Biosciences,TCG Lifesciences,Domainex,Absorption Systems,
What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00 , USD 6720.00, and USD 8960.00 respectively.
Are there any additional resources or data provided in the report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million .
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Preclinical Assets Analysis, Insights and Forecast, 2019-2031
- 5.1. Market Analysis, Insights and Forecast - by Type
- 5.1.1. Bioanalysis and DMPK Studies
- 5.1.2. Toxicology Testing
- 5.1.3. Compound Management
- 5.1.4. Safety Pharmacology
- 5.1.5. Others
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. North America
- 5.2.2. South America
- 5.2.3. Europe
- 5.2.4. Middle East & Africa
- 5.2.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Type
- 6. North America Preclinical Assets Analysis, Insights and Forecast, 2019-2031
- 6.1. Market Analysis, Insights and Forecast - by Type
- 6.1.1. Bioanalysis and DMPK Studies
- 6.1.2. Toxicology Testing
- 6.1.3. Compound Management
- 6.1.4. Safety Pharmacology
- 6.1.5. Others
- 6.1. Market Analysis, Insights and Forecast - by Type
- 7. South America Preclinical Assets Analysis, Insights and Forecast, 2019-2031
- 7.1. Market Analysis, Insights and Forecast - by Type
- 7.1.1. Bioanalysis and DMPK Studies
- 7.1.2. Toxicology Testing
- 7.1.3. Compound Management
- 7.1.4. Safety Pharmacology
- 7.1.5. Others
- 7.1. Market Analysis, Insights and Forecast - by Type
- 8. Europe Preclinical Assets Analysis, Insights and Forecast, 2019-2031
- 8.1. Market Analysis, Insights and Forecast - by Type
- 8.1.1. Bioanalysis and DMPK Studies
- 8.1.2. Toxicology Testing
- 8.1.3. Compound Management
- 8.1.4. Safety Pharmacology
- 8.1.5. Others
- 8.1. Market Analysis, Insights and Forecast - by Type
- 9. Middle East & Africa Preclinical Assets Analysis, Insights and Forecast, 2019-2031
- 9.1. Market Analysis, Insights and Forecast - by Type
- 9.1.1. Bioanalysis and DMPK Studies
- 9.1.2. Toxicology Testing
- 9.1.3. Compound Management
- 9.1.4. Safety Pharmacology
- 9.1.5. Others
- 9.1. Market Analysis, Insights and Forecast - by Type
- 10. Asia Pacific Preclinical Assets Analysis, Insights and Forecast, 2019-2031
- 10.1. Market Analysis, Insights and Forecast - by Type
- 10.1.1. Bioanalysis and DMPK Studies
- 10.1.2. Toxicology Testing
- 10.1.3. Compound Management
- 10.1.4. Safety Pharmacology
- 10.1.5. Others
- 10.1. Market Analysis, Insights and Forecast - by Type
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2024
- 11.2. Company Profiles
- 11.2.1 Eurofins Scientific
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Viroclinics Xplore
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 SGS
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Intertek Group
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 AmplifyBio
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 IQVIA
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 ICON plc
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Medpace
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Pharmatest Services
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 PPD
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Crown Bioscience
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Comparative Biosciences
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 TCG Lifesciences
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Domainex
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Absorption Systems
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.1 Eurofins Scientific
- Figure 1: Global Preclinical Assets Revenue Breakdown (million, %) by Region 2024 & 2032
- Figure 2: North America Preclinical Assets Revenue (million), by Type 2024 & 2032
- Figure 3: North America Preclinical Assets Revenue Share (%), by Type 2024 & 2032
- Figure 4: North America Preclinical Assets Revenue (million), by Country 2024 & 2032
- Figure 5: North America Preclinical Assets Revenue Share (%), by Country 2024 & 2032
- Figure 6: South America Preclinical Assets Revenue (million), by Type 2024 & 2032
- Figure 7: South America Preclinical Assets Revenue Share (%), by Type 2024 & 2032
- Figure 8: South America Preclinical Assets Revenue (million), by Country 2024 & 2032
- Figure 9: South America Preclinical Assets Revenue Share (%), by Country 2024 & 2032
- Figure 10: Europe Preclinical Assets Revenue (million), by Type 2024 & 2032
- Figure 11: Europe Preclinical Assets Revenue Share (%), by Type 2024 & 2032
- Figure 12: Europe Preclinical Assets Revenue (million), by Country 2024 & 2032
- Figure 13: Europe Preclinical Assets Revenue Share (%), by Country 2024 & 2032
- Figure 14: Middle East & Africa Preclinical Assets Revenue (million), by Type 2024 & 2032
- Figure 15: Middle East & Africa Preclinical Assets Revenue Share (%), by Type 2024 & 2032
- Figure 16: Middle East & Africa Preclinical Assets Revenue (million), by Country 2024 & 2032
- Figure 17: Middle East & Africa Preclinical Assets Revenue Share (%), by Country 2024 & 2032
- Figure 18: Asia Pacific Preclinical Assets Revenue (million), by Type 2024 & 2032
- Figure 19: Asia Pacific Preclinical Assets Revenue Share (%), by Type 2024 & 2032
- Figure 20: Asia Pacific Preclinical Assets Revenue (million), by Country 2024 & 2032
- Figure 21: Asia Pacific Preclinical Assets Revenue Share (%), by Country 2024 & 2032
- Table 1: Global Preclinical Assets Revenue million Forecast, by Region 2019 & 2032
- Table 2: Global Preclinical Assets Revenue million Forecast, by Type 2019 & 2032
- Table 3: Global Preclinical Assets Revenue million Forecast, by Region 2019 & 2032
- Table 4: Global Preclinical Assets Revenue million Forecast, by Type 2019 & 2032
- Table 5: Global Preclinical Assets Revenue million Forecast, by Country 2019 & 2032
- Table 6: United States Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 7: Canada Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 8: Mexico Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 9: Global Preclinical Assets Revenue million Forecast, by Type 2019 & 2032
- Table 10: Global Preclinical Assets Revenue million Forecast, by Country 2019 & 2032
- Table 11: Brazil Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 12: Argentina Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 13: Rest of South America Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 14: Global Preclinical Assets Revenue million Forecast, by Type 2019 & 2032
- Table 15: Global Preclinical Assets Revenue million Forecast, by Country 2019 & 2032
- Table 16: United Kingdom Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 17: Germany Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 18: France Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 19: Italy Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 20: Spain Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 21: Russia Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 22: Benelux Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 23: Nordics Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 24: Rest of Europe Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 25: Global Preclinical Assets Revenue million Forecast, by Type 2019 & 2032
- Table 26: Global Preclinical Assets Revenue million Forecast, by Country 2019 & 2032
- Table 27: Turkey Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 28: Israel Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 29: GCC Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 30: North Africa Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 31: South Africa Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 32: Rest of Middle East & Africa Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 33: Global Preclinical Assets Revenue million Forecast, by Type 2019 & 2032
- Table 34: Global Preclinical Assets Revenue million Forecast, by Country 2019 & 2032
- Table 35: China Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 36: India Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 37: Japan Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 38: South Korea Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 39: ASEAN Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 40: Oceania Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
- Table 41: Rest of Asia Pacific Preclinical Assets Revenue (million) Forecast, by Application 2019 & 2032
| Aspects | Details |
|---|---|
| Study Period | 2019-2033 |
| Base Year | 2024 |
| Estimated Year | 2025 |
| Forecast Period | 2025-2033 |
| Historical Period | 2019-2024 |
| Growth Rate | CAGR of XX% from 2019-2033 |
| Segmentation |
|
STEP 1 - Identification of Relevant Samples Size from Population Database



STEP 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note* : In applicable scenarios
STEP 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

STEP 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
Related Reports
About Market Research Forecast
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.

